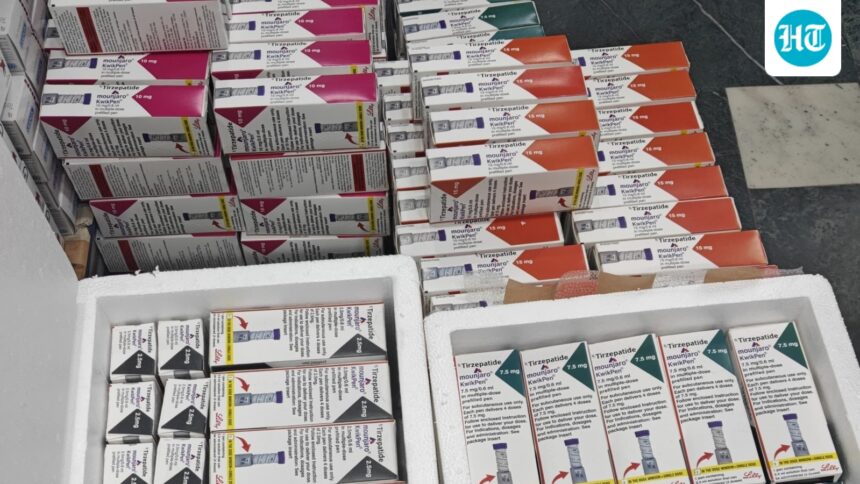
CHANDIGARH: In a late-night operation, the Haryana Food and Drug Administration (FDA), in coordination with the police, busted an alleged racket involving ‘fake’ Mongaro Quik Pen injections and arrested two persons after intercepting a vehicle in Gurugram.

According to the European Medicines Agency, Mongaro, a drug manufactured by an American multinational pharmaceutical company, is used to control type 2 diabetes in adults. It is also used to help people lose weight or keep it under control.
FDA officials said they acted after a report about the illegal sale and stockpiling of Monjaro injections in multiple doses (2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, and 15 mg).
According to the FDA memorandum, a team led by Narcotics Control Officer (DCO), Gurugram-1, Amandeep Chauhan, and police personnel from Sector 29 Police Station, Gurugram, laid a trap near HUDA City Center Metro station.
The US Food and Drug Administration said that at approximately 7:25 p.m., a vehicle suspected of carrying the shipment was intercepted. The occupants of the place were questioned in the presence of the team after independent witnesses refused to join the proceedings.
The driver, Shehbaz Alam Ansari, told officials that he was an Ola taxi driver posted in Delhi $1000 to drop a passenger in Gurugram. The passenger, identified as Jameel Khan, a Gurugram-based medical representative, was found carrying the injections.
DCO Amandeep Chauhan said that during interrogation, Majammil Khan said that the stock belonged to a person named Avi Sharma, a resident of Gurugram.
Sharma was contacted and he reached the spot around 12:10 am on April 19. However, he failed to provide any valid license to store, sell or distribute the medicines. He allegedly admitted that he was solely responsible for the consignment and revealed that the products were “manufactured” by him at his residence in Sector 62, Gurugram.
Both Khan and Sharma were arrested.
Market value
The FDA team seized the entire stock and sampled it according to procedure. The total value of the recovered syringes, based on the maximum retail price, was estimated at approx $The Coordination and Coordination Office said: 56.15 thousand.
The seized inventory included multiple batches of the Mounjaro KwikPen across all six strengths.
Officials also pointed out that the drugs were not stored under the specified temperature conditions of 2 to 8 degrees Celsius, raising further concerns about their safety and authenticity.
What the labels showed
Initial verification was done by comparing the seized products with the original label details shared by the manufacturer, Eli Lilly and Company (India) Pvt Ltd. Officials found multiple differences, including differences in font size, punctuation, formatting of manufacturing details, and toll-free numbers; But further investigation is still needed to determine whether the seized drugs are being trafficked and stored illegally or are completely counterfeit.
An official said the discrepancies indicate that the recalled stock is counterfeit and not manufactured by the original company.
Based on the findings, both Khan and Avi Sharma were immediately arrested for violations under Sections 18(C) and 17B of the Drugs and Cosmetics Act, 1940, Chouhan said.
These crimes are punishable under Article 27 of the law, which deals with the manufacture and sale of counterfeit drugs.
Officials said further investigations are underway to trace the supply chain, identify potential buyers and determine the extent of distribution of the suspected counterfeit syringes.